  Making weapons-grade uranium is very difficult and expensive, which is one reason so few countries have nuclear weapons. 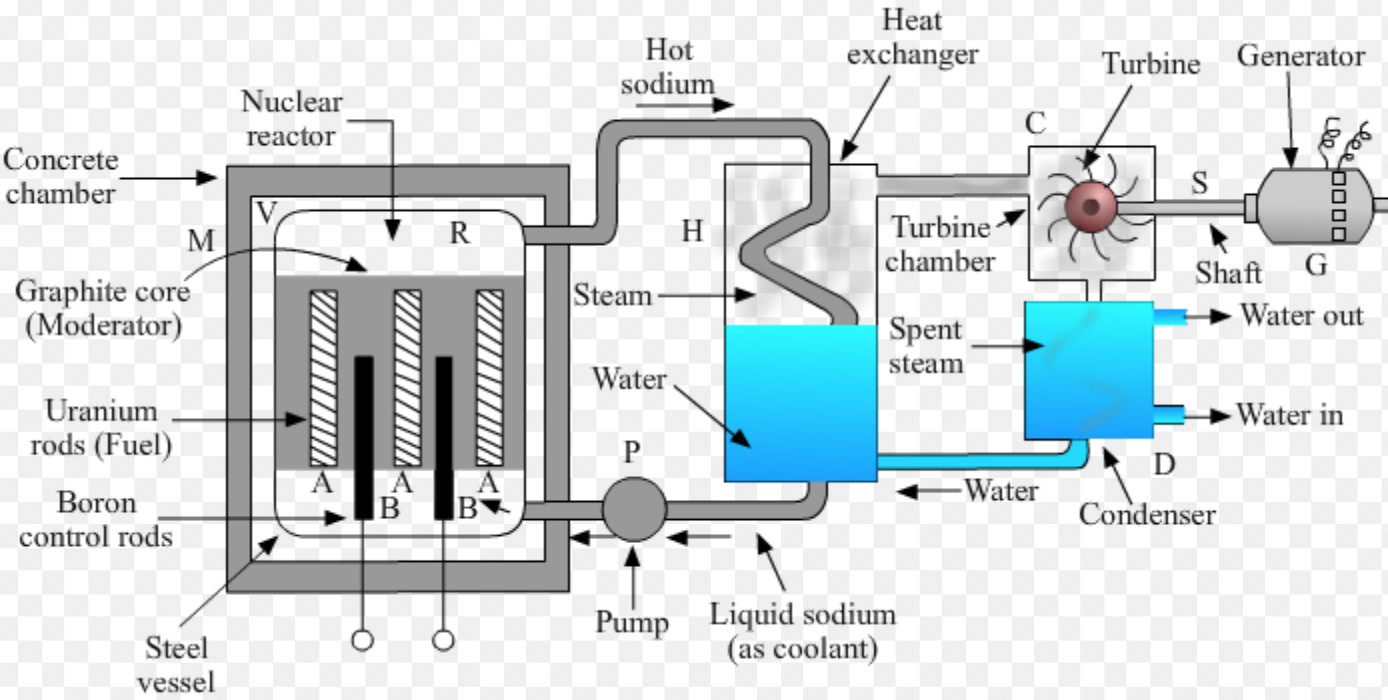
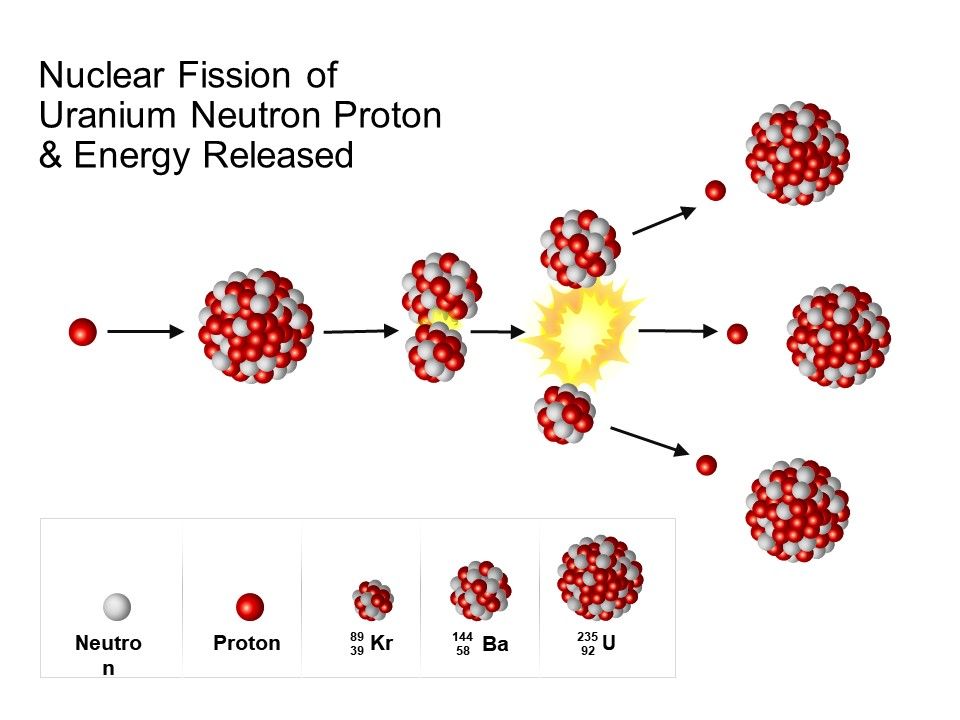
At first, there is only a slight increase in the concentration of U-235 atoms, so the process has to be repeated several times in the centrifuge to increase the enrichment. The force of the centrifuge separates the U-235 atoms from the U-238 atoms. The process of enriching uranium is done via a centrifuge after a gas has been created from the uranium. Three percent enrichment is sufficient for nuclear power plants, but weapons-grade uranium is composed of at least 90 percent U-235. īut for all of this to work, scientists have to first enrich a sample of uranium so that it contains 2 to 3 percent more U-235. The two atoms that result from the fission later release beta radiation (superfast electrons) and gamma radiation of their own, too. The splitting of an atom releases heat and gamma radiation, or radiation made of high-energy photons. That may not seem like much, but there are lots of uranium atoms in a pound (0.45 kilogram) of uranium The decay of a single U-235 atom releases approximately 200 MeV (million electron volts). The process of capturing the neutron and splitting happens very quickly. As soon as the nucleus captures the neutron, it splits into two lighter atoms and throws off two or three new neutrons (the number of ejected neutrons depends on how the U-235 atom splits). Uranium-235 (U-235) is only found in about 0.7 percent of uranium found naturally, but it is well-suited for producing nuclear power. Uranium undergoes spontaneous fission at a very slow rate, and emits radiation. The animation above shows a uranium-235 nucleus with a neutron approaching from the top. Nuclear fission occurs when one atom splits into two, creating energy. NUCLEAR FISSION URANIUM FUEL FREEFire a free neutron into a U-235 nucleus and the nucleus will absorb the neutron, become unstable and split immediately. It's also one of the few elements that can undergo induced fission. U-235 decays naturally by alpha radiation: It throws off an alpha particle, or two neutrons and two protons bound together. While there are several varieties of uranium, uranium-235 (U-235) is the one most important to the production of both nuclear power and nuclear bombs. Three types of moderators are used at the MIT reactor: (1) ordinary or "light" water that is also used to cool the reactor core, (2) deuterated or heavy water (D 20), and (3) high-purity graphite, both of which are excellent at slowing neutrons without absorbing them.Uranium is a common element on Earth and has existed since the planet formed. Since U-235 nuclei do not readily absorb the high energy neutrons that are emitted during fission, it is necessary to slow the neutrons down with a "moderator". In the MIT reactor, one other group of components is essential to the maintaining and controlling a chain reaction. As fewer and fewer neutrons are absorbed, more and more neutrons are available to cause the splitting of uranium nuclei, until finally enough neutrons are available to sustain a chain reaction. To put the reactor into operation, the control blades are raised very slowly. When the control blades are fully inserted, they absorb so many neutrons from the uranium that there are not enough to allow a chain reaction to continue. Boron has the property of absorbing neutrons without re-emitting any. The rate of fissions in the uranium nuclei in the MIT reactor is controlled chiefly by six control blades of boron-stainless steel which are inserted vertically alongside the fuel elements. When it is in operation, the central active core contains a huge number of neutrons traveling in every direction at very high speeds. The MIT Research Reactor is used primarily for the production of neutrons. If the country so chooses, it can secretly enrich the uranium to create weapons-grade uranium and harvest plutonium from uranium fuel rods for use in nuclear. Hence, the possibility exists for creating a chain reaction. Each time a U-235 nucleus splits, it releases two or three neutrons. Heat is created when nuclear fuel undergoes nuclear fission. This process is known as fission (see diagram below). Nuclear fuel is material used in nuclear power stations to produce heat to power turbines. When a U-235 nucleus absorbs an extra neutron, it quickly breaks into two parts. The arrangement of particles within uranium-235 is somewhat unstable and the nucleus can disintegrate if it is excited by an outside source. In the nucleus of each atom of uranium-235 (U-235) are 92 protons and 143 neutrons, for a total of 235. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
